Cancer treatments that work with the immune system are making great strides, with Keytruda from Merck being perhaps the best known. Still, there is much work to be done – both in developing unprecedented new treatments, as well as treatments that might combine with our existing treatments to make them even more effective. Triumvera (try-um-VEERA) Immunologics is working on both. Dr. Paul Lammers is its CEO.
Author: Staff
Triumvira Immunologics to Present Clinical Data from Phase I/II Trial TACTIC-2 Investigating TAC01-HER2 at the 2023 ASCO Annual Meeting
AUSTIN, Texas and HAMILTON, ON and SOUTH SAN FRANCISCO, Calif., May 16, 2023 /PRNewswire/ — Triumvira Immunologics, a clinical-stage company developing novel, targeted autologous and allogeneic T cell therapeutics that co-opt the natural biology of T cells to treat patients with solid tumors, today announced that the Company will be presenting clinical data on its lead asset TAC01-HER2 for the treatment of human epidermal growth factor receptor 2 (HER2) positive solid tumors at the 2023 American Society Of Clinical Oncology (ASCO) Annual Meeting, taking place in Chicago, IL, June 2-6. The presentation by Benjamin L. Schlechter, MD, from Harvard Medical School and a Senior Physician in Gastrointestinal Oncology at the Dana-Farber Cancer Institute, will highlight updated clinical data from the ongoing Phase I/II trial of TAC01-HER2 (NCT04727151) in patients with relapsed or refractory solid tumors.
“We are excited to present the latest clinical data from our Phase I/II trial of TAC01-HER2 in refractory solid tumors at the upcoming ASCO meeting as a poster and a poster discussion. The availability of updated clinical data will offer remarkable insights into the potential of TAC01-HER2 as a targeted therapy for patients who are refractory to existing HER2 targeted therapies,” said Deyaa Adib, M.D., Chief Medical Officer of Triumvira Immunologics. “We are committed to delivering innovative solutions for cancer patients and addressing critical unmet medical needs and look forward to sharing the updated findings with the oncology community at this prestigious scientific meeting.”
Joy and Boldness in Drug Development – with Paul Lammers
Join us in this episode as we talk with Paul Lammers, CEO of Triumvira Immunologics, a clinical-stage biopharmaceutical company focusing on developing T cell therapies for cancer treatment. Born in the Netherlands and now an American citizen, Lammers’ early life shaped his views on being bold and making connections with people a priority. He shares his insights on crucial but often neglected topics in life science leadership, while also revealing his joy as a father to three grown daughters and a grandfather.
We discuss the importance of transparent and open communication between leaders and employees, especially during times of change or uncertainty. Lammers also shares his thoughts on the challenges of balancing innovation and risk with regulatory and financial constraints. Additionally, we delve into the significance of investing in talent development and creating opportunities for employees to learn and grow, both for their own advancement and the success of the organization. Tune in to learn from Paul Lammers’ experiences and gain insights into effective leadership strategies in the biotech industry.
Triumvira Immunologics Presents Promising Data on TAC T Cells for the Treatment of HER2-overexpressing Solid Tumors at the 2023 AACR Annual Meeting
AUSTIN, Texas and HAMILTON, ON and SOUTH SAN FRANCISCO, Calif., April 18, 2023 /PRNewswire/ — Triumvira Immunologics, a clinical-stage company developing novel, targeted autologous and allogeneic T cell therapeutics, today announced the details of the Company’s presentation at the 2023 AACR Annual Meeting on its lead asset, TAC01-HER2, for the treatment of human epidermal growth factor receptor 2 (HER2) positive solid tumors. The presentations will include updated clinical data from the ongoing TACTIC-2 trial of TAC01-HER2 (NCT04727151) in patients with solid tumors and preclinical data on an allogeneic HER2-TAC T cell product.
Presentation details:
Title: A Phase I/II Trial Investigating Safety and Efficacy of Autologous TAC01-HER2 in Relapsed or Refractory Solid Tumors
Authors: Ecaterina E. Dumbrava, MD, Daniel Olson, MD, Samuel Saibil, MD, Brooke Pieke, Mridula A. George, MD, Riemke Bouvier, Kelly Gruber, Kara Moss, Nathan Ternus, Maria Apostolopoulou, Deyaa Adib, MD, and Benjamin L. Schlechter, MD.
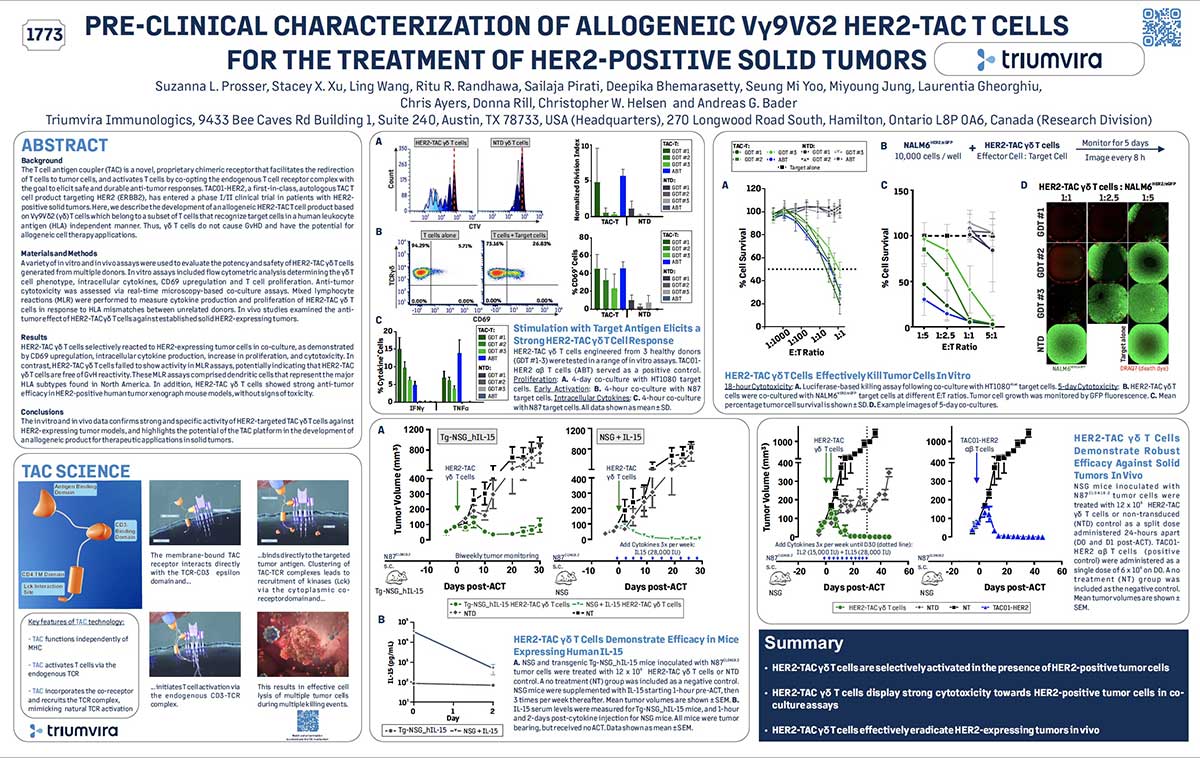
A phase I/II trial investigating safety and efficacy of autologous TAC01-HER2 in relapsed or refractory solid tumors
INTRODUCTION
• The T cell antigen coupler (TAC) is a novel, proprietary chimeric receptor that facilitates the re-direction of T cells to tumor cells and activates T cells by co-opting the endogenous T cell receptor complex, with the goal to elicit a safe and durable anti-tumor response. In preclinical models, TAC-engineered T cells effectively eradicate tumor cells in vitro and in vivo without toxicities typically associated with engineered T cell products. TAC01-HER2 is an autologous T-cell product comprising T cells expressing the HER2 TAC, which specifically recognize HER2+ cells.
• TACTIC-2 (NCT04727151) is an open-label, multicenter phase I/II study that aims to establish safety, maximum tolerated dose (MTD), recommended phase 2 dose (RP2D), pharmacokinetic profile, and efficacy of TAC01-HER2 in patients with HER2-positive solid tumors by immunohistochemistry that are 1+, 2+, or 3+ (i.e. breast, lung, pancreatic, colorectal, gastric, endometrial, ovarian, and others) whom have progressed on prior anti-cancer therapies.
• We present updated preliminary data from Cohorts 1-4 (19 participants) that highlights safety and efficacy data; the study further elucidates potential therapeutic impact to patients with HER2 overexpressed solid tumors.
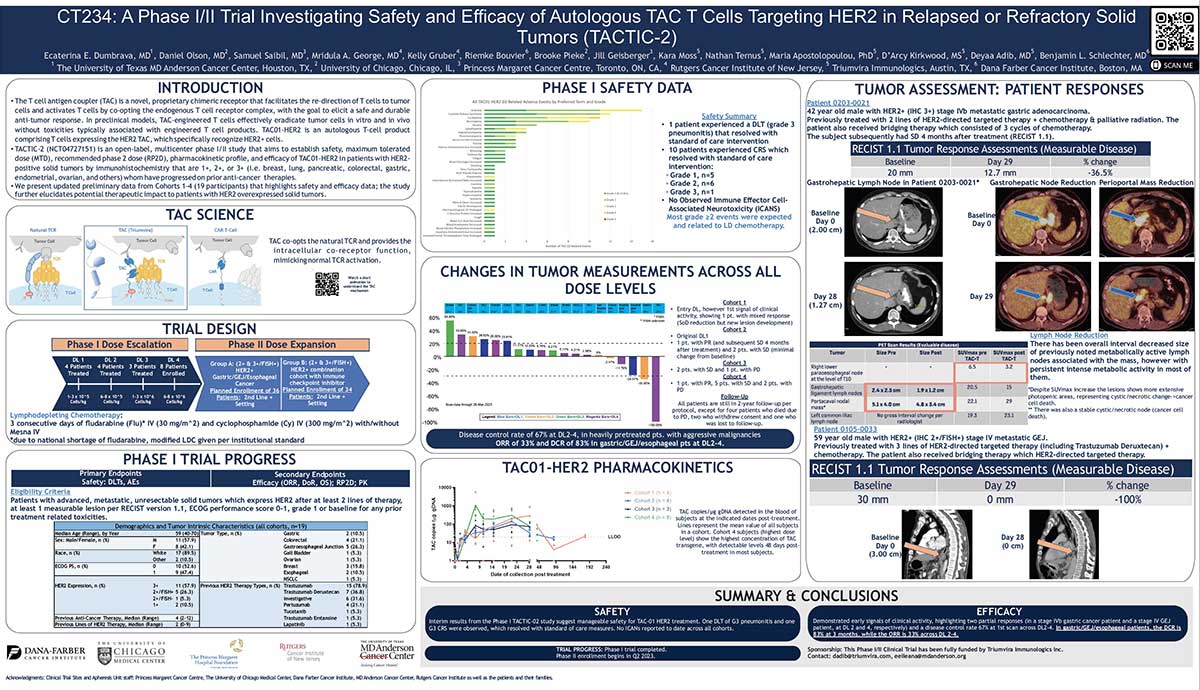
Patient-derived TAC01-HER2 TAC T cells produced in Cocoon® Platform is highly functional in models of solid tumors
Background
The T cell antigen coupler (TAC) is a novel, proprietary chimeric receptor that facilitates the redirection of T cells to tumor cells and activates T cells by co-opting the endogenous T cell receptor complex with the goal to elicit a safe and durable anti-tumor response. TAC01-HER2, a rst-inclass TAC T product targeting HER2 (ERBB2), has entered a Phase I/II clinical trial in patients with HER2-positive solid tumors. Here, we characterized the TAC T cell phenotypes and anti-tumor activity of TAC01-HER2 manufactured using leukocytes from Phase I/II patients in nonclinical in vitro and in vivo assays.
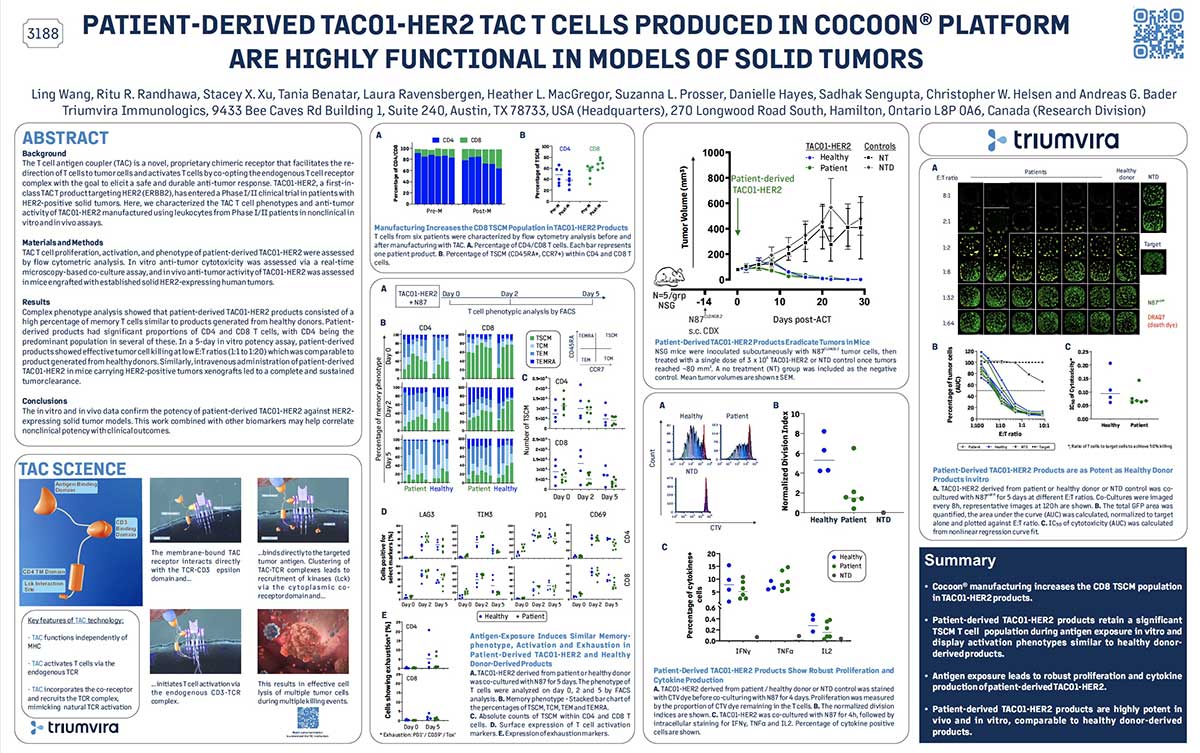
Preclinical characterization of allogeneic Vγ9Vδ2 HER2-TAC T cells for the treatment of HER2-positive solid tumors
Background
The T cell antigen coupler (TAC) is a novel, proprietary chimeric receptor that facilitates the redirection of T cells to tumor cells, and activates T cells by co-opting the endogenous T cell receptor complex with the goal to elicit safe and durable anti-tumor responses. TAC01-HER2, a rst-in-class, autologous TAC T cell product targeting HER2 (ERBB2), has entered a phase I/II clinical trial in patients with HER2-positive solid tumors. Here, we describe the development of an allogeneic HER2-TAC T cell product based on Vγ9Vδ2 (γδ) T cells which belong to a subset of T cells that recognize target cells in a human leukocyte antigen (HLA) independent manner. Thus, γδ T cells do not cause GvHD and have the potential for allogeneic cell therapy applications.
Engineering Better Cell Therapies for Cancers
Cell therapies are revolutionizing the way cancers are treated, but existing cell therapies have their limits. They have been more successful at treating hematologic tumors than solid tumors, and they can sometimes cause serious side effects, such as the destruction of antibodies or what’s known as cytokine storms in which the immune system gets over-revved and attacks healthy cells. Triumvira Immunologics is developing autologous and allogeneic T-Cell therapies that it believes can address the limitations of existing cell therapies and be used to treat both liquid and solid tumors.
We spoke to Paul Lammers, CEO of Triumvira, about the company’s platform technology, why it’s robust and versatile, and why its lead indication is for cancer where effective treatments already exist.
First in Human Episode #25 featuring Dr. Paul Lammers
For episode 25, we sit down with Dr. Paul Lammers, CEO of Triumvira Immunologics. Stay tuned to learn why having multiple programs in clinical trials can provide a safety net should one program fail and how it adds additional value for investors. First In Human is a biotech-focused podcast that interviews industry leaders and investors to learn about their journey to in-human clinical trials.
Presented by Vial, a tech-enabled CRO, hosted by Simon Burns, CEO & Co-Founder & guest host Co-Founder, Andrew Brackin.